Why is Gas Safety Important?

Unsecured liquid nitrogen dewar falls over and leaks at a university in Australia
Gas safety has always been important to labs - it’s the foundation of keeping your lab personnel safe when they are working. However, different methods of supplying gas to your applications offer different levels of safety.
Gas cylinders have long been used in labs around the world, but there are clear safety concerns when it comes to using these in your lab. Storage methods, handling and high pressures of compressed gases can all have devastating effects if an accident happens.
There have been a number of high-profile cases in the scientific world of accidents caused by gas leaks and explosions which have resulted in minor injuries, amputations, and, in some cases, loss of life.
This is why gas safety is so important to consider. Every employer should want to make sure their staff make it home safe.
What are the risks of stored gas?
The risks of using compressed gas cylinders for your stored gas range from minor injury to devastating results. The most obvious risk is the volume of gas that can be stored in each cylinder. Coupled with this is the high pressure that they are typically stored at. Should a leak occur in a gas cylinder, it would not take much of the capacity of the cylinder to render the lab a dangerous space for your staff.
With this in mind it is important that cylinders are stored in a well ventilated area with good accessibility and that they are fitted securely with the correct pipework which will need to be updated with every cylinder change.
Cylinders are also heavy and cumbersome which can result in personal injury if staff do not handle them correctly or, if dropped, result in a gas leak or act as a rocket propelling through the lab.
 With a gas leak in the lab, depending on the gas you use, you could find a significant oxygen depletion with a nitrogen leak or, with hydrogen, you could find you reach the Lower Explosive Limit of the gas.
With a gas leak in the lab, depending on the gas you use, you could find a significant oxygen depletion with a nitrogen leak or, with hydrogen, you could find you reach the Lower Explosive Limit of the gas.
You can read about how a gas generator helped avert disaster in Singapore back in 2020 here.
What Happens if the Oxygen Level Gets Too Low in my Lab?
Nitrogen cylinders and dewars are able to store huge volumes of pressurized nitrogen; a 50 liter gas cylinder contains 9000 liters of nitrogen at 200 bar pressure (the equivalent to 648 party balloons) and a 50 liter dewar contains approximately 34,800 liters of nitrogen (the equivalent to a fuel tanker).
Should an accident happen in your lab and a leak develops in your cylinder or dewar, you can expect the oxygen level in your lab to decrease significantly and, at these pressures, fairly quickly.
So what could you expect to happen if the oxygen level in your lab dropped?
| O2 (Volume %) | Effects and Symptoms |
|---|---|
| 18 – 21% | No discernible symptoms can be detected by the individual. A risk assessment must be undertaken to understand the causes and determine whether it is safe to continue working |
| 11 – 18% | Reduction of physical and intellectual performance without the sufferer being aware. |
| 8 – 11% | Possibility of fainting within a few minutes without prior warning. Risk of death when oxygen level drops below 11%. |
| 6 – 8% | Fainting occurs after a short time. Resuscitation possible if carried out immediately. |
| 0 – 6% | Fainting will occur almost immediately. Brain damage is likely, even if rescued. |
What is Lower Explosive Limit (LEL)?
The Lower Explosive Limit (LEL) is the lowest concentration of a gas or vapor that will burn in the air. The LEL varies from gas to gas, but for most flammable gases it is less than 5% by volume.
With regards to hydrogen, the LEL is only 4.1%. This means that a lab measuring 5m x 4m x 2.5m has an air volume of 50,000 liters. Given the LEL of hydrogen is 4.1%, it would only take 2,050 liters of hydrogen to reach this. If your lab is using a typical 50 liter hydrogen cylinder, containing roughly 9,000 liters of hydrogen, it would only need to release 25% of it’s contents to reach LEL.
Why is a Gas Generator Safer Than Cylinders?
A gas generator comes with a number of benefits and improved safety is just one of these benefits.
PEAK’s gas generators only supply gas on-demand, meaning your instruments only receive the gas they require for the application at hand. Our generators never store an amount of gas that would be unsafe in the event of a leak.
When we think about the previous examples of cylinder leaks and the dangers associated with these, a PEAK nitrogen gas generator contains less than 50 liters of gas at less than 8.5 bar pressure while our Precision Hydrogen generator produces up to 500cc/min of hydrogen. In comparison to the 9,000 liters of gas stored in cylinders, a gas generator significantly removes the risks associated with each gas usage.
With respect to our hydrogen generators, it would take almost three days to reach the LEL of hydrogen if a leak occurred – and this is if no hydrogen escaped from the lab in that time. However, the internal safety features in our generators would alert you to this leak before it could reach a level that should be of concern to any lab personnel.
How Does a Nitrogen Generator Work?
Nitrogen generators tend to fall into two technologies; a membrane generator or a Pressure Swing Adsorption (PSA) generator.
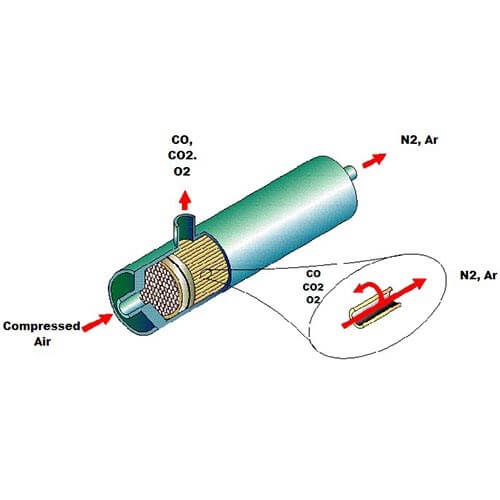
Illustration of the membrane inside some PEAK Nitrogen Generators
In a membrane generator, we force compressed air through a membrane - a bundle of small, hollow fibers. These are permeable to oxygen, but not to nitrogen. Nitrogen can pass through the end of the membrane bundle whereas the oxygen is purged out of the side which gives us the separation.
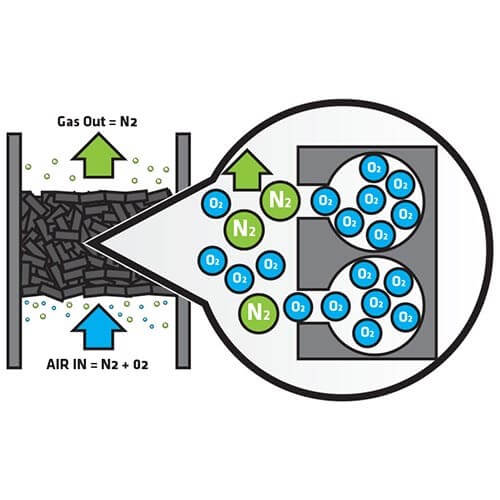
Illustration of the process of Pressure Swing Adsorption
With PSA nitrogen generators, we have a column which we pressurize and, as we pressurize the Carbon Molecular Sieve (CMS) material in the column, it will bind the oxygen from the compressed air allowing nitrogen to pass through to a buffer tank. We then have a depressurization phase where we are expelling the oxygen and, by continually pressurizing and depressurizing compressed air that’s coming into the system, we are able to build up purified nitrogen – typically less than 10ppm (sometimes less than 5ppm) of oxygen.
How Does a Hydrogen Generator Work?
A hydrogen generator is, effectively, a reverse fuel cell.
Rather than combining oxygen and hydrogen to create electricity, we’re taking electricity, providing this to a proton exchange membrane cell and using that to split water into oxygen and hydrogen. The proton exchange membrane is impermeable to oxygen and ions, but is permeable to hydrogen ions, which means that the hydrogen can pass through the cell whereas the oxygen can’t.

A selection of PEAK Hydrogen Gas Generators
What we are able to do, by applying a charge to the cell and separating the water, is purify hydrogen on one side of the cell and expel oxygen from the other.
What built-in safety features do PEAK generators have?
There are a number of safety features that are built into each of our PEAK generators to give you and your lab staff peace of mind as they work. These include:
- Internal leak check on start up
- External leak detection
- Pressure monitoring to prevent leaks and pressure build up
- Automatic cell shutdown at high pressure
- Audible/visual alarms
What Can a Gas Generator Be Used For?
PEAK have developed a range of high-quality gas generators tailored to the specific demands of a number of laboratory applications including LC-MS, GC, Sample Preparation, ICP-MS, FT-IR, and many more.
With dedicated solutions for a number of instrument manufacturers, PEAK is sure to have a generator suited to your needs
So if you are looking to improve your lab safety and make sure everyone gets home safe every day, PEAK are here to help.
