The aim within the laboratory should be to achieve the best separation in the shortest time period. The most commonly used gases as carrier gas for GC are nitrogen, hydrogen and helium.
The differences between the gases are evident when comparing their van Deemter curves. This is illustrated in the Van Deemter Equation (Figure 1).
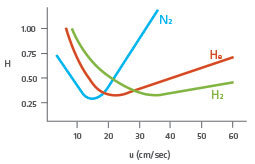
Figure 1: The Van Deemter Equation
Although it displays the lowest minimum plate height compared with that of helium or hydrogen, nitrogen has a much narrower velocity range and a steeper van Deemter curve, so at higher flow rates, solute efficiency drops off dramatically (Figure 1). Analysis times with helium are about 1/2 the value when using nitrogen and there is only a very small sacrifice in efficiency. The helium Van Deemter curve is much flatter than the nitrogen curve, thus changes in the average linear velocity do not decrease efficiency by a large amount. Hydrogen is the fastest carrier gas (uopt), with an optimum linear velocity of 40cm/sec, and exhibits the flattest Van Deemter profile. Hydrogen’s high uopt (optimal linear velocity) results in the shortest analysis times. Also, the wide range over which high efficiency is obtained makes hydrogen the best carrier gas for samples containing compounds that elute over a wide temperature range.
Although Nitrogen, Helium and Hydrogen can all be considered suitable carrier gases for use in GC, historically helium has been the most widely used due to the safety concerns associated with hydrogen and also the fact that nitrogen is much less efficient!
Why Change from Helium
The emerging helium shortage means that people have no choice but to look at other alternatives. Are we running out of Helium completely? No, not quite yet but a shortage of helium and increased demand within the medical, scientific and industrial fields is leading to this rare commodity rising in price. Many leading GC Manufacturers understand the impact this may have in the laboratory and have started to actively recommend switching your carrier gas from Helium to Hydrogen.
There are major benefits of using Hydrogen as your carrier gas:
- Increased speed: increasing the linear flow rate allows for shorter run times, thereby increasing the throughput of your laboratory.
- Lower temperature separations: at the faster elution times, it may not be necessary to increase the column temperature run rate. It may even be possible to lower the maximum temperature needed for the analysis.
- Longer column life: lower temperatures lead to less column bleed, which in turn can mean a longer column life. In addition, hydrogen is a reducing gas and can remove potential acidic sites off the column, further increasing column life!
- Availability: Hydrogen is readily available through the electrolysis of water and with a Peak Gas Generator it can be generated on demand.
- Hydrogen gas is already being used in the laboratory for a variety of purposes: It is the fuel gas for the most commonly used detectors (FID) and therefore already present in most GC Labs.
The selection of an appropriate hydrogen supply source for GC contributes to the reliability of the system and results gained. Although hydrogen cylinders are still the most commonly used supply source in the laboratory, hydrogen generators are gaining in popularity. Understanding the benefits and drawbacks of each option helps in choosing the best hydrogen supply source for a system.
Hydrogen Cylinders Supply Concerns and Considerations
There are many safety concerns associated with the use of hydrogen cylinders including:
- Cylinder handling and storage - the frequent changing of cylinders interrupts the GC’s operation and can be time consuming and the system must be monitored to ensure that gas supply does not run out. Changing can also increase the risk of contamination of ambient air interfering with the analysis.
- Safety - The flammable nature of hydrogen and the high pressure of cylinders present concerns for cylinder the handling and storage within the lab. In addition, the size and weight of hydrogen cylinders present hazards to personnel performing cylinder changeouts. Care must be taken and cylinders should be secured to the wall or bench top using appropriate cylinder holders and restraints.
- Variation in Quality of the Gas - High purity hydrogen is required for use as a carrier gas to ensure a clean baseline. Contaminant levels for a given grade of hydrogen may vary from cylinder to cylinder, and still meet the specification. This cylinder-to-cylinder variation may be a consideration for sensitive analyses, as it introduces another variable into the operation.
- Selection of Appropriate Gas Delivery Equipment to Ensure System Purity - To minimise the chance of contaminant, such as oxygen and moisture, from entering the GC system during cylinder changes, high purity gas handling equipment should be used.
Hydrogen Generators as an Alternative to Cylinders
Safety also plays a major role in justifying the use of hydrogen generators within the laboratory, they provide a continuous source of high purity hydrogen and can eliminate many safety concerns over using hydrogen cylinders.
Increased Safety Using a Hydrogen Generator
Using a hydrogen generator has the following safety features:
- Hydrogen generation on demand. There is no storage of hydrogen and the generator output is typically low enough that the laboratory air can never build up to explosive levels.
- Eliminates the need for personnel to handle large quantities of the flammable, high-pressure gas.
- Automatic shut down on leak detection.
- Mechanical shut down fail safe.
- No risk of new leaks when changing of cylinders.
Using a hydrogen generator has the following benefits:
- No increased contamination when you get to the bottom of the cylinder
- No changing cylinders - time saving.
- Space saving as most hydrogen generators have a very small footprint.
- Based on cylinder gas savings alone, a hydrogen generator pays for itself in one or two years.
Conclusion
There are many considerations when choosing a suitable gas for GC. Using hydrogen as opposed to helium or nitrogen has many advantages and switching can be done successfully with minimal disruption to previous analysis. Additionally, using hydrogen allows for higher throughput in the lab and increased column life. Whilst hydrogen as a carrier gas is deemed dangerous when supplied via cylinders, many of these safety fears are eliminated when using a hydrogen generator. With the shortage and expense of helium, the switch to hydrogen can be a cost saving solution, particularly in view of increased productivity and prolonged column life.

Ed Connor DR.SC. is GC-MS Application Specialist, PEAK Scientific, Inchinnan Business Park, Scotland, UK . Prior to joining PEAK in February 2013, Ed completed his Dr.Sc. at ETH Zurich in Switzerland using GC-MS to look at herbivore induced plant volatiles and their interaction with beneficial insects. He then joined the University of Zurich where his work focused primarily on volatile collection methods and analyses using GC-MS and GC-FID . +44 141 812 8100, econnor@peakscientific.com
Find out more about PEAK Scientific's hydrogen, nitrogen and zero air Precision series generators for GC or contact us.